FAQ - Batteries
Much of this is reproduced with grateful acknowledgment from http://www.climber.org/gear/index.html.
The rest was added by the maintainer from contributions on aus.bushwalking.
Send any comments to the maintainer Roger Caffin
Index
Notes collected from the Climber.Org Gear List, which generally reached the consensus that 'it all depends on what you need'. [This index duplicates the one above, but in a different order - RNC.]
Few of us (me included) have ever taken more than one set of batteries and used them in a similar enough way to obtain a valid comparison. Hopefully the comments below will help you choose wisely!
Carrying spares is a function of battery size/weight/type
Richard Schwaninger wrote: I use the [Petzel] Zoom on longer trips and I always use the 4.5 V battery. It lasts... well, I don't quite know [how long] since I replace the battery before each trip.
Paul Wilson wrote: My old headlamp uses 2 AAA batteries. And I use fresh ones for trips that need a full charge. The used batteries go on the shelf and I use them for my mag light in my shop and the battery eating GPS. I seem to have a good supply of used batteries.
Steve Eckert wrote: That's the big problem with the big battery. I prefer smaller batteries that I don't mind carrying spares for. The big ones are so heavy I've almost never seen anyone carry a spare, but I have seen a couple of DEAD big batteries because the headlamp turned on in a pack and ran all day. Rhetorically, what do you do with all the 1/4-used battery packs? I switched from expensive to cheap alkalines because I always carry spares and I almost never run the first set down. The cheap ones are lighter, so this way I'm carrying less weight. That's not going to be reflected in ANY manufacturer spec!
Craig Smith wrote: I always keep a set of regular lithiums in my pack as an emergency backup. These will last for years w/o self-discharging, and are nice to use if your rechargeable batteries run down before the end of the day.
GPS (etc) battery meter shows remaining life incorrectly for NiMH
Kevin Craig wrote: I tried NiMH in my Garmin (eTrex's and 12XL). There is some kind of incompatibility between the battery meter algorithm in the Garmins and the 'power curve' or whatever it's called for NiMH batteries that causes them to always register as much less than 'full' even with fresh batteries right out of the charger. Since I like to know how much juice I really have left, I abandoned them in favor of the LiIon which seem to work OK.
Craig Smith wrote: Regarding batteries for the Garmin GPS: I have an eTrex and use rechargeable Nickel metal hydride batteries most of the time. Works fine with very reasonable battery life. As someone else mentioned, the output voltage of the NiMH batteries is lower than the alkaline or lithium for which the eTrex is designed, so the battery capacity bar graph will read
less than 'full' even with a newly charged NiMH battery set. But the life will be as good or better than a set of alkalines.
Rich Feldman wrote: An independent issue is the low-voltage cutoff of the equipment. NiMH drop to 1.2 volts early in the discharge, but hit 1.1 just before exhaustion. With alkalines, most of the energy is delivered above 1.25 volts, but you are throwing away a useful fraction if you stop at 1.2 or even at 1.1.
Longest-lasting battery is different in high vs. low current applications
Alan Ritter wrote: I use the NiMH batteries exclusively in my digicam (Nikon 990). It has been my experience with my Garmin GPS II+ that the NiMHs don't last as long in the GPS as regular alkalines, which is just the opposite of the digicam. It may have to do with the respective power supplies and what their minimum Vin requirements are.
Richard Vassar wrote: Battery capacity, especially for alkaline cells, is a function of the current drain from the load. In high current drain applications, e.g. flashlights, CD players, digital cameras, lithium AA cells have a significantly higher mAh capacity than alkaline batteries.
Kevin Craig wrote: Richard, thanks. That squares with my experience using lithiums in FRS radios, headlamps, GPS etc. all high-drain applications.
Rich Feldman wrote: Yeah, what Alan just said about NiMH outlasting alkalines in a digicam but the opposite in radio receivers! A while back I posted an explanation here, with URL's to battery technical data (e.g. under duracell.com, look for OEM information).
The Mallory charts give the AA size alkaline cell a capacity of about 2800 mAH, but that's at room temperature, under light loading, with end of life at about 1.0 or 0.9 volt. Under similar conditions the NiMH are around 1500 mAH as Alan says. But if you draw over 500 mA, like the Coolpix with monitor on, the alkaline really sucks, and dies after less than 900 mAH. This is where NiMH (and NiCd) excel: the ability to deliver most of their energy at relatively high currents (even the 1 hour rate is no sweat). (By the way, the coolpix is very
tolerant of battery voltage variations, in fact draws more current at lower voltage, which implies a switching-mode DC-DC converter).
I don't know how lithium cells stack up, but in the previous thread here, someone gave us a URL to tech data. At low current they outlast alkalines, and much more so at low temperature. Don't know about their fast-discharge performance, but many digicams now come with a starter set of lithium cells. (often each 3-volt cell is packaged to replace a side-by-side pair of AA's).
Paul Wilson wrote: The Lithiums are much better for high current draw as in a photoflash usage wherein the battery has a significant recovery time between flashes and rewinds. Alkalines perform best when subjected to low steady current draw, as in a gps or modern LED headlamp. All the reports I can find about Lithiums having longer life at normal temps are for usage in cameras or other intermittent, high current applications. Can anybody provide reports comparing low current draw between the two different batteries? Real
info would be better than the photo people testimonials. If someone has Backpacker from some time last year they compared Lithiums against alkaline's. Dont know about current draw or temperature, but I was told it was a low temperature test.
BTW, I would not consider my LED headlamp as a high current drain device with a alkaline battery life or about 20 hours. My gps has similar low drain characteristics. Even my old halogen lamp was good for 6-8 hours on good old low mAh alkalines.
Steve Eckert wrote: Battery capacity depends STRONGLY on temperature (which most people recognize) but ALSO on current drain (which few are aware of). Some alkaline batteries have a longer life in a clock than sitting on the shelf not providing current at all. The slow discharge keeps them from leaking. Other batteries appear to discharge completely when used in a bright (high-current) headlamp, but if you take them out for an hour or two they will magically 'recharge' and you can get more use out of them. Sort of like sopping water off the floor only to find that more water seeps out of the soaked walls. That's the kind of battery that would work better in a low-current application.
Those ratings like 3000mAh, mean '3 amp-hours'. That can TECHNICALLY be a 3 amp current provided for 1 hour, or 1 amp provided for 3 hours.... but good documentation requires keeping those two numbers separate. They will NOT be the same. You can't JUST look at the mAh rating. You've got to know whether it was tested with high or low current, intermittent or steady drain, and at what temperature.
Alan Ritter wrote: Steve is quite correct. If you go to the battery manufacturers' WWW sites (energizer.com for instance) and look up the technical data on batteries, you will find that alkaline batteries have vastly different energy content depending upon the drain to which they're subjected. By 'vastly', I mean 3:1 or more, getting worse and worse as the drain goes up. Alkalines are also more temperature sensitive than lithium cells. Part of this has to do with the internal impedance (resistance) of the battery, part to do with the chemistry. That's why alkalines are absolutely worthless in high-drain applications like digital cameras but work great in relatively low-drain applications like LED headlamps, Walkman radios (not CD
players), etc.
As usual, it's a matter of matching the tool to the task. I have a couple of small single-LED flashlights that run off 9V batteries. Using them fairly regularly (but briefly) on Scout outings, the original batteries lasted nearly a year. The one that really died would actually burn brighter on the 'low' setting than on 'high' at the very end. The internal resistance of the battery had gone up enough that the loss across it consumed enough power that the LED could not achieve its maximum brightness. I don't know how long the second set will
last...they're out of my 'change the battery in the smoke detector every six months' box, but, hey, if I get six months' use out of them, they're free...sort of...
Longest-lasting battery is different in moderate vs. low temperature applications
Paul Wilson wrote: For arctic expeditions most use a battery pack for the headlamp that is worn inside the clothing so they work fine at low ambient temps since the batteries do not get cold.
Alan Ritter wrote: My experience with the NiMH batteries has been in moderate temps, down to near freezing, but certainly not winter conditions. They seem to hold up pretty well down to 32F.
Kevin Craig wrote: NiMH's also suffer from the cold; to a lesser extent than alkalines but MUCH more than lithiums. A matter of fractions of ounces, but the Lithum batteries are also noticeably lighter than either alkalines or NiMH's.
[You will find that trying to use an alkaline battery in the snow, or below freezing, will give very poor life. Their internal chemistry dies. However, the Eveready 1.5 V Lithiums will work well under sub-zero conditions, and are recommended. True, they are distinctly dearer, but they last a lot longer on many things such as LED lights, and the extra cost is worth it. The 1.5 V AA size is readily available. The 1.5 V AAA size is not yet available (early 2004), but Eveready promises it is is coming later in the 2004 year. R N C]
Even in ideal conditions, capacity varies with battery type
Paul Wilson wrote: I just looked up alkaline capacities and the Eveready's best alkaline is rated at 3135mAh and their new Lithium Photo is listed at 2900mAh. This validates Monty's post. Who should we believe, Eveready or various testimonials? Consumers Reports tested alkaline's and said that the Duracell Ultra is significantly better than the Eveready. The cost at Walmart is 0.95 vs 2.48 each,less for the alkalines when on sale. Lithiums seem to be pretty hard to find in the out of the way places I tend to go. Not many Walmarts or camera shops.
Another observation: The Lithiums at 2900mAh and weigh 0.51 oz, Eveready alkaline 3135mAh, 0.81 oz and Duracell Ultra, 0.95 oz (all per battery). Or 7647mAh/Oz, and 3870mAh/Oz. A huge difference.
Don't confuse [lithium batteries in] this discussion with the [rechargeable] lithium-ion batteries that come in many electronic gadgets these days. They are not the same as the Eveready Photo lithium's.
Alan Ritter wrote: When you buy NiMH batteries, be careful of capacities (printed on the battery). The older Energizer AA NiMHs were only 1200 mAH and my Nikon 990 eats them awfully fast. Radio Shack has 1600 mAH AA cells. The latest Energizers (blue and gold color scheme) are 1600 mAH, but I haven't tried them yet. Thomas has some 1700 mAH AA cells, but they're pricey...
Ed Lulofs wrote: On a per weight basis, [lithium batteries] have 4 times as much energy per gram [as standard alkalines]. So I only use lithium batteries when backpacking. I gave up on my Tikka because AAAs are not available in lithium [yet]. I think that lithium [batteries] and LEDs are the most weight efficient and reliable light source.
Craig Smith wrote: The capacity of NiMH batteries has been slowly increasing. The best ones you can buy in most stores now are 1500 or 1600 mA hours. But if you go to the internet or some camera stores, look for batteries branded as either MaHa or Quest. These are available in 1700 and 1800 mA hours for AA size. Quest also makes a very nice charger for them that will charge 2 or 4 AAs or AAAs with either AC or car battery. I highly recommend these products - have used them extensively with both GPS and digital photo gear.
Battery Life (summarized from battery technical info taken from Eveready's website)
Monty Smith wrote: In prepping for Denali, I wanted to find out how different batteries performed in the cold, both for storage as well as power output. I found much good info at Eveready's website, and the following is a summary of the tables and graphs.
Summary:
- Lithium batteries are by far the best for cold usage. The voltage stays constant as they discharge and they have the lowest temperature storage and usage ranges. They are more expensive, however.
- Despite theories to the opposite, storing a battery in the cold will not discharge it, and in fact cold storage is good for batteries. They need to be warmed for proper power output, however.
General notes on all batteries:
Batteries described here in detail are all non-rechargeable 1.5V AA batteries, since that's what commonly used in our equipment, although there's a quick mention of rechargeables at the end. Storage for all batteries is down to -40F, but operating range varies widely, as does performance at various temperatures - only cold temperature info is provided. Although % storage capacity loss per year (shelf life) varies widely by both temperature and battery type, if being stored at 0C the worst battery (carbon-zinc) only loses 3% per year, so cold storage while on a climb won't affect any battery's capacity, but will affect its power output. Internal resistance, or the ability to maintain constant voltage as the power demand increases, is generally low (constant voltage) for all types except carbon-zinc. Power output varies from 950mAh to over 3000mAh.
Definitions:
- Shelf life: I've chosen to use the number of years for the battery to drain by 10%. Shelf life for all batteries is doubled or tripled if stored in the freezer, so the shelf life at 0C will be used.
- mAh: milliamp-hours. The number of hours the battery can sustain a 1-milliamp drain and is a measure of how much power the battery has.
- Sloping or flat discharge curve: whether the battery's voltage decreases as the power drains, or if it's flat. Flat is generally better, but also means you can have less warning that the battery's wearing out. [However, while a flat voltage characteristic is often nice, it can be detrimental to the lifetime of Maglite bulbs and even white LEDs, if the circuit design assumed a significant internal impedance in the battery. R N C]
- Carbon-zinc: These are the plain, old-fashioned batteries, and have power of 950mAh with a sloping discharge curve. Operating range is only down to 20degF and shelf life is 1/3 to 1/10th of other types - about 3.5 years. They perform very poorly at low temperature: at -5degF their service life is decreased by 75% and output drops quickly as the temperature drops below room temperature. [I found they died at 0 C. R N C] On the plus side, they're cheap.
- Alkaline: The Energizer has 2850mAh and the new Energizer e2 has about 15% more, or 3135mAh, and both have a sloping discharge curve. Operating range is down to 0degF and shelf life is 10+ years. They're not much better at low temperature; at -5degF their service life is decreased by 60% and output drops quickly as the temperature drops below room temperature. Although they're more expensive, they're comparable to carbon-zinc in terms of cost per hour of use.
- Silver oxide: These are the small (dime-shaped) batteries often used in cameras and calculators. Since they don't come in AA size, comparative power output is meaningless, but they have a flat discharge curve. Operating range is down to 14degF and shelf life is 10+ years. They're a better at low temperature; at -5degF their service life is decreased by 50%. Output decreases slowly until about 40degF but below that output drops quickly.
- Lithium: Lithium AAs won't outlast alkaline, with equivalent power of 2900mAh but with a flat discharge curve. Operating range is down to -40degF and shelf life is 10+ years. They're significantly better at low temperature; at -5degF their service life only is decreased by 20% and output decreases slowly as the temperature drops. They're also much more expensive. [Distinguish between the two sorts of Lithium batteries avaiable: there are some giving 1.5 V and other giving 3 V. don't mix them! R N C]
- Rechargable batteries: NiCads and Nickel-metal-hydrides have less storage capacity - only half that of alkalines, they supply lower voltage, and also drain much faster by themselves. They do not perform well at low temperatures, roughly equivalent to carbon-zinc.
Richard Booth wrote:
I have shifted to NiMH for most applications in order to cut down on the disposing of batteries. They work well in cameras, headlamps, gps, etc. Just a personal preference.
Alkaline: Primary cell (not rechargeable). These have good shelf life, that is they can be left around for years and still maintain good capacity. Poor high current drain applications such as digital cameras but ok for headlamps. Poor low temperature performance. Checking the capacity specifications should reveal the rate, in a fraction of the capacity, that they can be discharged. Usually .1C or for a 2500 mahour battery this would be 250 ma for about ten hours at room temperature. Typical capacities are 2500 mahour.
NiMH: Nickle metal hydride. Secondary cell (re-chargeable). These have poor shelf life and will supposedly discharge in some months. I use these in a digital camera and they work well for months in this application going through hundreds of pictures. A set of alakalines will barely make it to about 40 pictures. NiMH are initially expensive but if re-charged about ten times the cost is approximately the same as alkaline. The weight of the NiMH batteries is about the same as alakaline. Typical high performance cells run about 1700 mAhour which is less than alkaline. My NiMH batteries in my digital camera worked ok at -17F winter ice climbing. NiMH is supposedly better than alkaline at cold temperatures but the curves only modestly support this. One observation: get NiMH from a reputable source. They are sold by all sorts of bogus types. Try Thomas Distributing [UK dealer] for Maha batteries and charger [or
AA Battery Endurance Testing
Paul Wilson wrote: We all use AA batteries for our gadgets such as head lamps, GPS receivers, radios and the like. The typical alkaline batteries have been the most popular since they are readily available and can be found discounted at many big box stores. Recently, Eveready came out with new chemistry, 'Lithium/iron Disulfide (Li/FeS2)'. The proper name for this battery is 'Eveready Photo Lithium'. This new battery comes from the store with an initial voltage of 1.775 volts average compared to the Duracell Ultra of 1.61 volts average. This portends that the lithium battery will perform better due to higher potential. Lithium batteries are also well known for their low temperature and long shelf life as well as much higher cost.
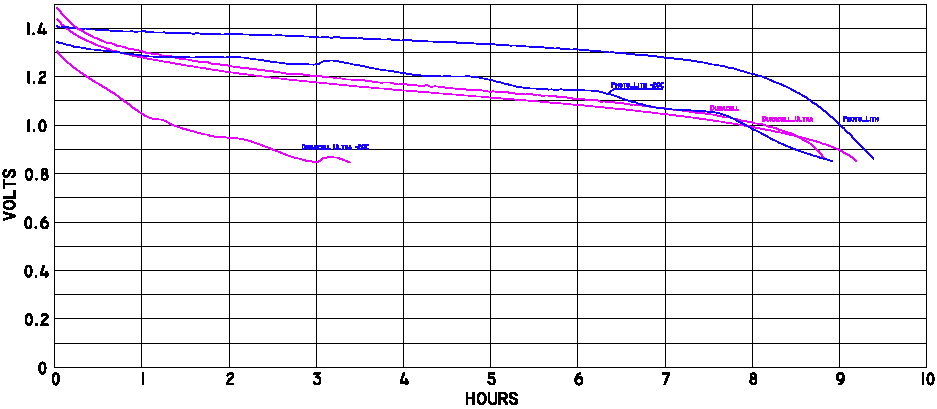
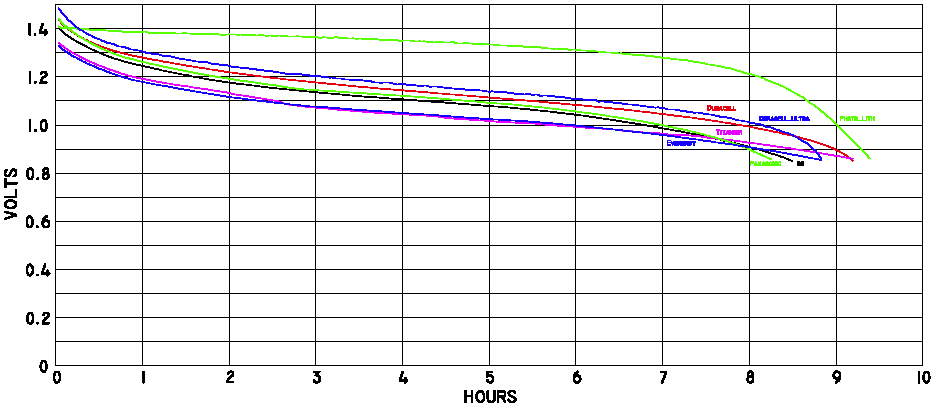
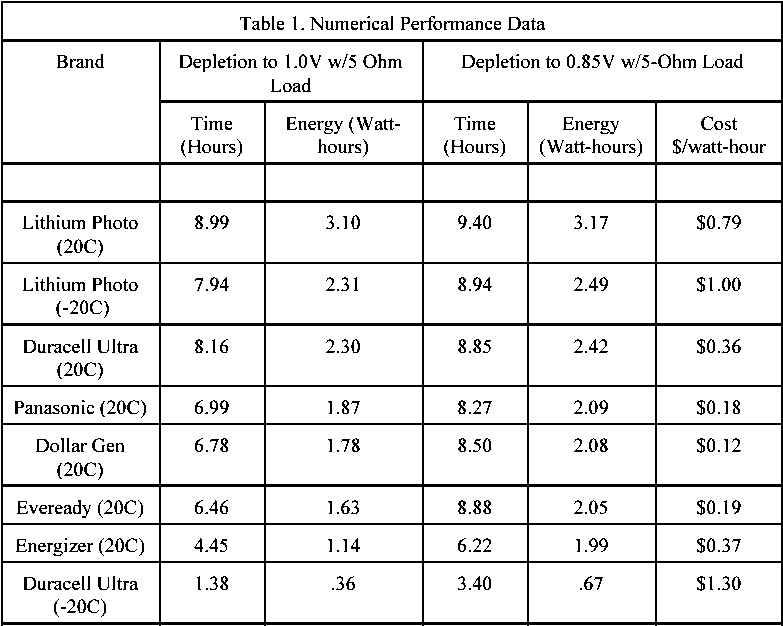
In the sport plane field the subject came up as pilots often use the same stuff climbers use. So, one of the electrical experts decided to test a bunch of batteries. I provided him with the extra batteries so low temperature data could be collected. This person is Bob Nuckolls III. What he did was write software for his laptop and wire up the batteries to allow them to be discharged through a 5 ohm load each to a level of 1.0 and 0.8 volts at room temperature and in his freezer. Let's examine the numerical data in table 1 which resulted from the testing.
Standard alkaline battery comparisons at 20°C: The Duracell Ultra has the greatest energy at 2.42 watt-hours, but the Dollar General batteries have the lowest cost, $0.12 per battery. So the discount house gives good value but not as long a life compared to the best alkaline battery which is the Duracell Ultra.
Lithium battery comparisons at 20°C: The lithium battery compared to the Duracell Ultra has 31% more energy and is 120.% $/watt-hour higher in cost. The lithium battery compared to the Dollar General has 52% more energy and is 558% $/watt-hour higher in cost.
Comparisons at -20°C: The Lithium has 272% more energy than the Duracell and costs 23% $/watt-hour LOWER in cost.
Conclusions: If the temperature is real cold, use lithium instead of Duracell Ultra for lower cost and for longer life. At normal
temperatures and long trips select Duracell Ultra. For shorter trips select the cheapest alkaline batteries you can get and change them as required.
| Click here for a scalable PDF version of this table (from the climber.org web site).
The real numbers from the computer are as follows: The lithium took 30,500 seconds to drop to 0.9 volts at -20°C and it ran 33,680 seconds at room temperature. The Duracell Ultra poops out at 8,800 seconds at -20°C and it runs 31,430 seconds at room temperature.
Notes:
- Most devices (except lights) are designed to operate down to about .8 to 1.0 volts per cell. Of course if the climber has switched to the LED headlamps then the time to discharge the batteries is so long at normal temperature the cheapest battery would last for many alpine starts. But for cold high altitude climbs I would choose the lithium's.
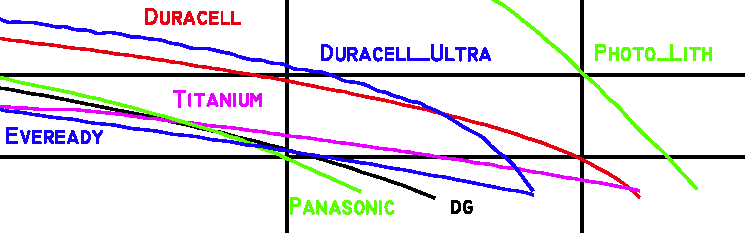
- The plots below are quite revealing since if one knows the drop dead voltage on the device the battery life can be predicted. The curves show different characteristics near 0.9 volts at normal temperatures so the Duracell Ultra would last much longer that any of the Eveready alkalines. Although I chose not to clutter up the plots with the other brands I found it was interesting that the discount store house brands fall between the two mentioned. I just purchased 48 Kirkland brand cells for $12 at Costco but these were not tested. The package said 'Made in the USA/Fabrique Aux E-U' for what it is worth.
|
 |
Memory Effect in Batteries
The memory effect is denied by some on the rather odd contention that the "real" Ni-chemistry memory effect is something that can only be seen under special laboratory conditions; or else in some exotic application like early communications satellite batteries that were charged and discharged in a regular, consistent way as the thing moved in and out of eclipse. True, but hardly relevant...
Practically, there is a different, but very real effect, which manifests as an obvious loss of capacity in Ni batteries that are regularly recharged from a partially discharged state. I call that "memory effect", as most people do.
This arises from the progressive growth of coarser-grained oxyhydroxide crystals in the active cell material with each charge/discharge cycle. Charging following partial discharge deposits extra material onto crystals that have not been completely consumed by the partial discharge - the leftovers act like seed crystals in a child's "crystal growth" experiment. Larger crystals have less surface area per unit volume, and so are less reactive. They tend to be preferentially preserved over charge/discharge cycles, and go on growing bigger and more numerous with time.
These less-reactive large crystals cause the cell voltage to fall faster during discharge. When the voltage falls below some cut-off voltage, we say the battery is "flat". With lots of large crystals, that happens well before all the active material is consumed. So the capacity appears to be (well, "is") degraded... [Incidentally, this process can occur regardless of the use of a smart or dumb charger.]
This effect can be severe for NiCads, where it happens at both electrodes. It is less severe for NiMH, where it occurs at the positive electrode only. Ignore marketing hype that says that NiMH has "no memory effect".
Fortunately, the effect is completely reversible and easily preventable. Occasionally (say every dozen or so cycles) make sure you discharge the battery to nearly* flat, before recharging fully. Severely "memory affected" cells may need a few such deep discharge cycles in a row for full recovery.
*the "nearly" there is CRITICALLY important.
Single cells can be completely flattened at a suitable rate without harm, but a "battery" (several cells in series, like in your Petzl) must not be completely flattened. Say you just left your Petzl on overnight to fully flatten the batteries - what happens is that the weakest cell in the thing runs flat first, and is then charged in reverse as the remaining cells run down. (Think about it...) It is that "reverse current" effect that does the damage - instant cell death if it is prolonged and severe. For the same reason, you must not mix up charged and flat cells in the one device!
Best to run your torch until there is the barest glimmer from the low beam, then remove the cells.
From "Glen F" , Friday, 2 April 2004
Battery chargers
Humbug recommended http://www.web-ee.com/primers/files/fsect5.pdf for a good reference on battery charging. It is a pdf file, so you will have to download it (from here or there). I have read it, and I thought it was very good.